Fueiceel® Research Grade 25cm2 Electrolyzer to Convert CO2 to HCOOH is a precision-engineered device designed for the electrochemical conversion of CO2 into formic acid. This system utilizes a three-compartment design to optimize the conversion process. In the cathode compartment, humidified CO2 is electrochemically reduced to formate anions, while in the anode compartment, deionized (DI) water is electrolyzed to generate protons. The formate anions pass through an anion exchange membrane into the central compartment, while protons are transferred through a cation exchange membrane into the same compartment. The combination of formate anions with protons in this central compartment results in the formation of formic acid, which is collected at the outlet.
This 25 cm² electrolyzer includes a titanium anode flow field, a central compartment with ion exchange media, a stainless steel cathode flow field and all necessary accessories such as nuts, bolts, o-rings, gaskets, and an insulating kit.
A complete electrolyzer with ion exchange media (in central compartment), catalyst-coated electrodes, Nafion® and Sustainion® membranes are also available upon request. The complete electrolyzer is fully assembled, tested, and verified to ensure optimal performance before shipment.
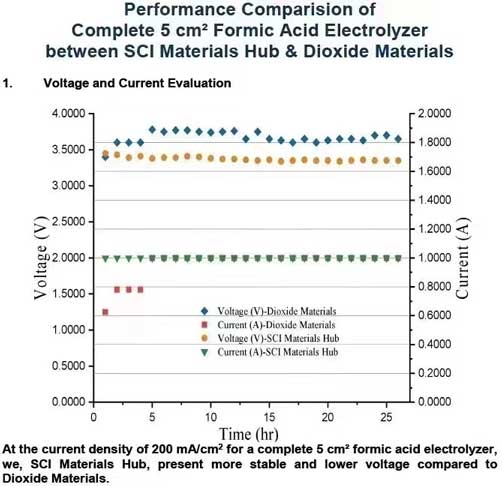
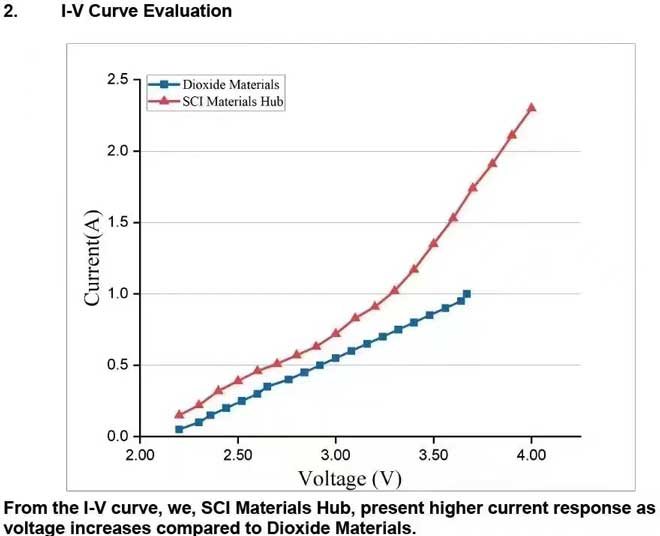
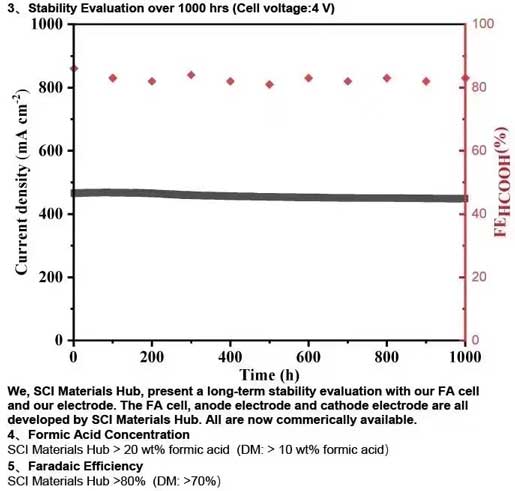
At the current density of 200 mA/cm² for a complete 5 cm² formic acid electrolyzer, we, SCI Materials Hub, present more stable and lower voltage compared to Dioxide Materials. We, SCI Materials Hub, present a long-term stability evaluation with our FA cell and our electrode. The FA cell, anode electrode and cathode electrode are all developed by SCI Materials Hub. All are now commerically available.
Formic Acid Concentration SCI Materials Hub > 20 wt% formic acid (DM: > 10 wt% formic acid)
System Setup
To set up the system, begin by connecting a peristaltic pump to circulate DI water from a reservoir to the anode chamber at a flow rate of 3 mL/min. A second peristaltic pump should feed DI water to the central compartment at a suggested flow rate of 0.065 mL/min through the inlets located on the backside of the anode flow field. Adjusting the flow rate into the central compartment can vary the concentration of produced formic acid—typically, a slower flow rate favors the production of higher concentration formic acid, albeit with decreased Faradaic efficiency. It is recommended to use 1/8” OD, 1/16” ID PTFE tubing for all connections. Pure CO2 from a cylinder should be humidified using a bottle humidifier (sold separately) and then fed to the cathode chamber at a flow rate of 30 sccm.
Tubing Connections
Cathode: Remove the nuts from the compression fittings and extract the black rubber rods from the nuts. Insert the 1/8” OD PTFE tubing through the nuts, and then secure the nuts onto the compression fittings. Connect the tubing from the CO2 humidifier to the compression fitting at the top of the cathode (stainless steel flow field) and tighten the nuts by hand. Connect another piece of PTFE tubing to the bottom compression fitting on the cathode and route it to the catholyte collector, and then to the exhaust. Since CO is poisonous and H2 is flammable, ensure that the cathode gas product is not released into the lab or working area.
Anode: Remove the nuts from all compression fittings and the black rubber rods from the nuts. Insert the 1/8” OD PTFE tubing through the nuts, and secure the nuts onto the compression fittings. Follow the labels on the compression fittings to connect the tubing accordingly, and tighten the nuts by hand.
Power Connections
Locate the threaded hole for the wire connection on the top of the cell. Attach the ring terminal with a Phillips round head screw. Repeat this procedure for both the anode and cathode connections.
Cell Testing and Operation
To begin testing, start the DI water flow from the reservoir to the inlet of the anode chamber at a rate of 3 mL/min, and to the central compartment at a flow rate of 0.065 mL/min. Feed humidified CO2 into the cathode chamber at a flow rate of 30 sccm. Connect the anode electrical lead (red) and cathode electrical lead (black) to the positive and negative terminals, respectively, on the power supply. Set the initial voltage to 4.5-5.0V and the current to 2.5A (0.1A/cm²). As the voltage stabilizes over time, gradually increase the current to 1.5A, 2A, and then 2.5A. The electrolyzer should reach stable operating conditions within several hours, depending on membrane and electrode conditioning.
For long-term testing, it is recommended to perform a reverse polarity treatment at 1.5V for 30 seconds approximately every 100 hours to maintain cell performance.